Understanding Skids in Pharmaceutical Manufacturing
In the complex landscape of pharmaceutical manufacturing, the term “skid” might not immediately ring a bell for everyone. Nevertheless, it plays a pivotal role behind the scenes, serving as an indispensable component in the production of life-saving medications. Skids, in the context of pharmaceutical manufacturing, refer to modular and compact systems designed to streamline and optimize various stages of the pharmaceutical production process. These systems, in particular, are meticulously engineered to handle critical tasks such as material handling, mixing, filtration, and more. As a result, skids are the unsung heroes of pharmaceutical manufacturing, ensuring precision and efficiency in the creation of drugs that improve and save countless lives.
Importance of Skids in Pharmaceutical Production
Pharmaceutical production is a highly regulated and exacting field, where the quality and consistency of products are paramount. To meet these demands, skids are instrumental in maintaining these standards. Moreover, they offer pharmaceutical manufacturers a level of control, precision, and adaptability that is unparalleled. From the careful handling of raw materials to the intricacies of fermentation and purification, skids are versatile tools that contribute to the reliability and safety of pharmaceuticals. Not only that, their importance extends beyond just production efficiency; skids also play a crucial role in reducing waste, minimizing contamination risks, and ensuring compliance with stringent regulatory requirements.
Purpose and Scope of the Article
The purpose of this article is to demystify the world of skids in pharmaceutical manufacturing. Specifically, we will delve into the intricacies of how skids are integrated into different stages of pharmaceutical production and the critical role they play in ensuring product quality and consistency. In addition, we will explore the key components and design considerations when implementing skid systems.
The Role of Skids in Pharmaceutical Manufacturing
Overview of Pharmaceutical Manufacturing Process
In order to understand the significance of skids in pharmaceutical manufacturing, it’s essential to have a broad overview of the pharmaceutical manufacturing process. This process involves a series of steps aimed at converting raw materials into finished pharmaceutical products that are safe, effective, and compliant with regulatory standards. Key stages in this process include raw material handling, mixing and blending, fermentation and bioprocessing, filtration and separation, purification and crystallization, and finally, formulation and packaging.
Integration of Skids in Different Stages of Manufacturing
Raw Material Handling
- Skids play a pivotal role in the initial stages of pharmaceutical manufacturing by facilitating the efficient handling of raw materials. Specifically, this includes the transportation, storage, and precise measurement of ingredients, ensuring accuracy and consistency in the composition of pharmaceutical products.
Mixing and Blending
- Next, the homogenous mixing of various components is crucial to pharmaceutical production. Skids equipped with specialized mixing and blending systems ensure that ingredients are uniformly distributed, which is especially important for the quality and efficacy of the final product.
Fermentation and Bioprocessing
- In addition, in biopharmaceutical manufacturing, skids are employed in fermentation processes where microorganisms or cells are used to produce therapeutic proteins or other biologics. They are designed to control critical parameters such as temperature, pH, and agitation, maintaining optimal conditions for biological growth and product yield.
Filtration and Separation
- Furthermore, skids are essential for the filtration and separation of solids and liquids during the manufacturing process. They enable precise control over filtration parameters, thereby ensuring the removal of impurities and unwanted particles from the product stream.
Purification and Crystallization
- During the subsequent stage, purification and crystallization, skids assist in the isolation and purification of the active pharmaceutical ingredient (API). They help in maintaining the necessary temperature, pressure, and solvent conditions for the purification process, thus ensuring high-quality API production.
Formulation and Packaging
- Finally, in the last stages of pharmaceutical manufacturing, skids are used to prepare the formulated drug product and package it into its final form, such as tablets, capsules, or vials. By doing so, these skids streamline the formulation process and ensure accurate dosing, ultimately leading to consistent product quality.
Advantages of Skids in Pharmaceutical Production
The integration of skids into various stages of pharmaceutical manufacturing offers several significant advantages:
- Efficiency: Skids automate and streamline many critical processes, reducing manual labor and minimizing the risk of human error, resulting in increased overall production efficiency.
- Consistency: Skids provide precise control over process parameters, ensuring product uniformity and batch-to-batch consistency, which is crucial for regulatory compliance and product quality.
- Flexibility: Skids can be designed for scalability, allowing pharmaceutical manufacturers to adapt to changing production demands and accommodate different batch sizes.
- Compliance: Skids are designed with compliance in mind, helping pharmaceutical companies adhere to strict regulatory requirements and quality standards, such as Good Manufacturing Practices (GMP).
- Cost-effectiveness: By optimizing processes and reducing resource wastage, skids contribute to cost savings over time, making pharmaceutical manufacturing more economically viable.
- Quality Assurance: Skids offer real-time monitoring and data logging capabilities, enabling manufacturers to track and document the entire production process, ensuring the highest quality standards are met.
- Competitive Advantage: Companies that adopt skid-based manufacturing processes often gain a competitive edge in the pharmaceutical industry by delivering high-quality products efficiently and at a competitive price point.
The Role of Skids in Pharmaceutical Manufacturing
Overview of the Pharmaceutical Manufacturing Process
To understand the significance of skids in pharmaceutical manufacturing, first and foremost, it’s essential to have a broad overview of the pharmaceutical manufacturing process. In general, this process involves a series of steps aimed at converting raw materials into finished pharmaceutical products that are safe, effective, and compliant with regulatory standards. Throughout these stages, key operations include raw material handling, mixing and blending, fermentation and bioprocessing, filtration and separation, purification and crystallization, and finally, formulation and packaging.
Integration of Skids in Different Stages of Manufacturing
Raw Material Handling
- Skids play a pivotal role in the initial stages of pharmaceutical manufacturing by facilitating the efficient handling of raw materials. This includes the transportation, storage, and precise measurement of ingredients, ensuring accuracy and consistency in the composition of pharmaceutical products.
Mixing and Blending
- The homogenous mixing of various components is crucial to pharmaceutical production. Skids equipped with specialized mixing and blending systems ensure that ingredients are uniformly distributed, which is vital for the quality and efficacy of the final product.
Fermentation and Bioprocessing
- In biopharmaceutical manufacturing, skids are employed in fermentation processes where microorganisms or cells are used to produce therapeutic proteins or other biologics. Skids control critical parameters such as temperature, pH, and agitation, maintaining optimal conditions for biological growth and product yield.
Filtration and Separation
- Skids are essential for the filtration and separation of solids and liquids during the manufacturing process. They enable precise control over filtration parameters, ensuring the removal of impurities and unwanted particles from the product stream.
Purification and Crystallization
- During purification and crystallization, skids assist in the isolation and purification of the active pharmaceutical ingredient (API). They help in maintaining the necessary temperature, pressure, and solvent conditions for the purification process, ensuring high-quality API production.
Formulation and Packaging
- In the final stages of pharmaceutical manufacturing, skids are used to prepare the formulated drug product and package it into its final form, such as tablets, capsules, or vials. These skids streamline the formulation process and ensure accurate dosing, leading to consistent product quality.
Advantages of Skids in Pharmaceutical Production
The integration of skids into various stages of pharmaceutical manufacturing offers several significant advantages:
- Efficiency: Skids automate and streamline many critical processes, reducing manual labor and minimizing the risk of human error, resulting in increased overall production efficiency.
- Consistency: Skids provide precise control over process parameters, ensuring product uniformity and batch-to-batch consistency, which is crucial for regulatory compliance and product quality.
- Flexibility: Skids can be designed for scalability, allowing pharmaceutical manufacturers to adapt to changing production demands and accommodate different batch sizes.
- Compliance: Skids are designed with compliance in mind, helping pharmaceutical companies adhere to strict regulatory requirements and quality standards, such as Good Manufacturing Practices (GMP).
- Cost-effectiveness: By optimizing processes and reducing resource wastage, skids contribute to cost savings over time, making pharmaceutical manufacturing more economically viable.
- Quality Assurance: Skids offer real-time monitoring and data logging capabilities, enabling manufacturers to track and document the entire production process, ensuring the highest quality standards are met.
- Competitive Advantage: Companies that adopt skid-based manufacturing processes often gain a competitive edge in the pharmaceutical industry by delivering high-quality products efficiently and at a competitive price point.
Components of a Pharmaceutical Manufacturing Skid
Core Components
Pumps
- Explanation of the role of pumps in pharmaceutical skids.
- Types of pumps commonly used in pharmaceutical manufacturing.
- Considerations for selecting the right pump for specific applications.
- Integration of pumps in the skid’s design for optimal performance.
Valves
- The significance of valves in regulating fluid flow within pharmaceutical skids.
- Types of valves utilized in pharmaceutical manufacturing skids.
- Valve materials and their compatibility with pharmaceutical processes.
- Precision control and automation of valves for process efficiency.
Instruments and Sensors
- Overview of instruments and sensors used for monitoring and control.
- Examples of sensors measuring variables like temperature, pressure, and flow rate.
- How instruments and sensors contribute to process optimization.
- Integration of real-time data from instruments and sensors into the skid’s control system.
Auxiliary Components
Control Systems
- Explanation of the central role of control systems in pharmaceutical skids.
- Types of control systems (e.g., PLC, SCADA) and their functionalities.
- Customization of control systems to meet specific manufacturing requirements.
- Safety measures and redundancy in control systems for pharmaceutical processes.
Heating and Cooling Systems
- Importance of temperature control in pharmaceutical manufacturing.
- Types of heating and cooling systems (e.g., heat exchangers) used in skids.
- Energy efficiency considerations in heating and cooling.
- Integration of heating and cooling systems into the skid’s overall design.
Data Logging and Monitoring
- The role of data logging in maintaining product quality and process control.
- Systems for data collection, storage, and retrieval.
- Importance of compliance with regulatory requirements for data monitoring.
- Real-time monitoring and alarm systems for prompt action in case of deviations.
Importance of Material Selection and Compatibility
- The critical significance of material selection in pharmaceutical skids.
- Materials commonly used in skid construction and their properties.
- Considerations for material compatibility with pharmaceutical products and processes.
- Ensuring the absence of contamination risks through material selection.
- Compliance with regulatory guidelines regarding material usage in pharmaceutical manufacturing.
Key Considerations in Skid Design and Integration
Customization vs. Standardization
In pharmaceutical manufacturing, the decision between customization and standardization of skids is crucial. Customized skids are designed to meet specific production needs and processes, offering maximum efficiency and control. However, they can be more expensive and time-consuming to develop. On the other hand, standardized skids are pre-designed and readily available, often more cost-effective, but may not perfectly align with unique manufacturing requirements. Striking the right balance between customization and standardization is essential to optimize the manufacturing process.
Regulatory Compliance and Documentation
Compliance with pharmaceutical regulations and documentation standards is non-negotiable. Skid design and integration must adhere to Good Manufacturing Practices (GMP), ensuring the production of safe and high-quality pharmaceuticals. Robust documentation, including validation reports, standard operating procedures (SOPs), and equipment qualification, is essential for regulatory audits and product quality assurance. Ensuring that skids meet regulatory requirements is paramount to avoid legal and quality control issues.
Scalability and Flexibility
Pharmaceutical manufacturing operations often require scalability and flexibility to adapt to changing production demands. Skid systems should be designed with the capability to scale up or down without major disruptions. Additionally, flexibility in skid design allows for accommodating different products and processes within the same facility. Modular skid systems with interchangeable components can be a strategic choice to achieve scalability and adaptability.
Maintenance and Upkeep
Regular maintenance is vital to ensure the longevity and reliability of skid systems. Consideration must be given to accessibility for maintenance tasks, ease of component replacement, and predictive maintenance strategies. Planned maintenance schedules should be established to minimize downtime and prevent unexpected breakdowns, which can disrupt pharmaceutical production schedules and impact product quality.
Cost Analysis and Budgeting
Cost analysis and budgeting are critical factors in skid design and integration. Pharmaceutical manufacturers must weigh the initial investment in skid systems against long-term operational cost savings and efficiency gains. It’s essential to evaluate the total cost of ownership, taking into account not only the purchase cost but also energy consumption, maintenance expenses, and potential production losses due to downtime. A thorough cost-benefit analysis helps make informed decisions about skid design and integration that align with the organization’s financial goals and operational efficiency.
These considerations play a pivotal role in ensuring that skid systems are not only optimized for pharmaceutical manufacturing but also contribute to overall business success by meeting regulatory standards, maintaining flexibility, and managing costs effectively.
Need a reliable partner?
Red River specializes in the design and manufacturing of pressure vessels. We also fabricate related items such as prefabricated spools and skid packages.
Reach Out to us today and experience the Red River difference. Where American Made and American Values come together, we care more.
FAQ: Skids in Pharmaceutical Manufacturing
What is a skid in pharmaceutical manufacturing?
A skid in pharmaceutical manufacturing refers to a process system or module mounted on a frame or platform, which can be easily transported and installed as a unit. Skids typically contain integrated equipment, piping, electrical systems, and instrumentation for specific manufacturing processes, such as formulation, filtration, or purification.
Why are skids used in pharmaceutical manufacturing?
Skids are used in pharmaceutical manufacturing for their flexibility, efficiency, and cost-effectiveness. They allow for pre-assembled and tested process units to be easily integrated into manufacturing facilities, reducing on-site installation time and minimizing disruptions. Skids also offer scalability and can be designed for easy expansion or reconfiguration as production needs change.
How do skids improve the pharmaceutical manufacturing process?
Skids improve the pharmaceutical manufacturing process by providing pre-engineered and pre-fabricated systems that are compact, easily transportable, and quick to install. This reduces project timelines, lowers labor costs, and ensures higher quality control through off-site fabrication. Skids also enable modular manufacturing, allowing pharmaceutical plants to rapidly adapt to new products or process changes with minimal downtime.
Can skids be customized for specific pharmaceutical processes?
Yes, skids can be fully customized for specific pharmaceutical processes. Manufacturers work closely with clients to design and build skids that meet unique process requirements, including the integration of specialized equipment, adherence to stringent hygiene and safety standards, and compliance with regulatory requirements. Customization ensures that each skid perfectly fits the intended application, optimizing performance and efficiency.
What types of equipment are commonly included on pharmaceutical skids?
Common types of equipment included on pharmaceutical skids encompass reactors, mixers, heat exchangers, pumps, filters, chromatography systems, and storage tanks. Instrumentation for monitoring and control, as well as necessary piping and electrical components, are also integrated into the skid design to provide a complete, ready-to-operate process unit.
Related Blog Post
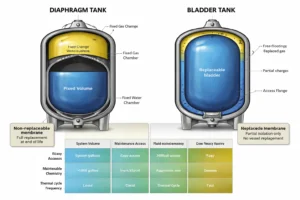
When to Choose Bladder vs Diaphragm Tanks
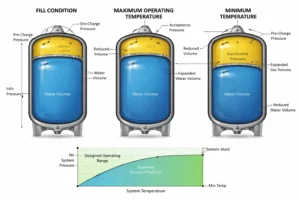
How Do Expansion Tanks Control Loop Pressure?

Expansion Tanks for Data Center HVAC

Which Tests Verify Buffer Tank Quality?

What Size Buffer Tank for Data Centers?
About Author