Molecular sieves are high-performance drying agents that remove trace water from solvents more efficiently than traditional methods. This guide covers how they work, which type to use (3A, 4A, 5A), and how to apply them safely and effectively in lab or industrial settings. Includes step-by-step instructions, regeneration tips, and real-world FAQs.
Molecular Sieves and Their Role in Solvent Drying
At Red River, we understand that drying solvents with precision is critical to maintaining consistent product quality and operational efficiency. Whether you’re in chemical manufacturing, petrochemical processing, or pharmaceuticals, using the right molecular sieves for solvent drying can prevent water contamination, protect your reactions, and meet strict industry standards. This guide walks you through how molecular sieves work, which types to use, and how to integrate them into your drying protocol.
What Are Molecular Sieves?
Molecular sieves are highly porous materials that capture molecules based on length and polarity. Imagine them as tiny, selective sponges that efficiently eliminate water and other undesirable molecules from your solvents. Unlike conventional drying agents, their functionality makes them essential tools within the molecular sieve solvent drying procedure. They consistently deliver steady results, ensuring your solvents are free from moisture and ready for important processes.
Why Use Molecular Sieves for Drying Solvents?
Even trace amounts of water can cause unwanted reactions, reduce solvent effectiveness, or degrade sensitive compounds. Molecular sieves remove this moisture at the molecular level, outperforming traditional agents like calcium chloride or silica gel in both speed and selectivity.
Key advantages:
- Removes water to <0.01% (100 ppm) in most solvents
- Regenerable via heating to 250°C
- Compatible with common solvents: THF, toluene, ethanol, etc.
- Works even at low temperatures or high humidity
Types of Molecular Sieves for Solvent Drying
Choosing the proper kind of molecular sieve is vital for achieving superior effects. Each molecular sieve targets a specific molecule, making them highly versatile. Commonly used types encompass 3A, 4A, and 5A molecular sieves, each providing unique advantages for solvent dehydration. For instance, 3A sieves are ideal for drying unsaturated hydrocarbons, while 4A and 5A sieves are suitable for general solvent drying and removing water from alcohols and gases. Understanding which type best suits your needs is key to maximizing efficiency.
| Type | Pore Size | Best For | Notes |
| 3A | 3 Å | Unsaturated hydrocarbons, ethanol | Excludes larger molecules to prevent reaction |
| 4A | 4 Å | General solvents, water from alcohols | Common default |
| 5A | 5 Å | CO₂ removal, polar solvents | Larger pore for small organics |
Step-by way of-Step Guide: How to Use Molecular Sieves to Dry Solvents
Preparing Your Solvent for Drying
Before adding molecular sieves, ensure your solvent is as clean as possible. Remove any impurities that could interfere with the drying process. Filtering the solvent beforehand is usually recommended to remove any particulate matter.
Choosing the Right Molecular Sieve
Select the right sieve based on solvent type and moisture content. For instance, 3A sieves excel at drying unsaturated hydrocarbons, while 4A or 5A sieves are preferred for general solvent drying.
Adding Molecular Sieves to the Solvent
Add the sieves to the solvent in a controlled environment. Use 10–20% of the sieve’s weight relative to the solvent. Ensure even distribution for optimal moisture removal, as explained in how to dry over molecular sieves.
Monitoring the Drying Process
Allow the solvent and molecular sieves to interact for some hours, or maybe overnight, depending on the solvent and moisture level. Stirring now and again can help speed up the technique.
How to Know When Your Solvent Is Fully Dried
Test moisture content using Karl Fischer titration. Once the desired dryness is achieved, remove the sieves via filtration.
Best Practices for Efficient Solvent Drying with Molecular Sieves
Tips for Maximizing Molecular Sieve Efficiency
- Choose the right type for your solvent; match pore size to molecular length.
- Keep sieves in an airtight container before use to prevent moisture absorption.
- Pre-activate sieves by heating to remove residual moisture.
- Use the correct ratio of sieves to solvent (10–20% by weight).
- Stir or agitate periodically during drying for improved results.
Common Mistakes to Avoid
- Using the wrong sieve type
- Overloading solvent with sieves, which may reduce efficiency
- Failing to pre-activate sieves
- Improper storage that allows moisture uptake before use
For advanced techniques, check ultimate guide to recharging dry desiccant.
Take Your Solvent Drying to the Next Level
Ensure consistent product quality and operational efficiency with the right molecular sieves.
Contact Red River today to explore our high-performance solutions, get expert guidance on selecting and applying sieves, and experience reliable, American-made equipment built for precision.
Frequently Asked Questions
1. What are the benefits of the use of molecular sieves over different drying retailers for solvents?
Molecular sieves provide numerous blessings over conventional drying agents like silica gel or anhydrous salts. First, molecular sieves have a particularly specific pore length that permits them to selectively adsorb water molecules whilst leaving the solvent molecules unaffected. This selectivity makes them distinctly efficient at putting off moisture, even at low concentrations. Additionally, molecular sieves can be regenerated and reused in multiple instances, making them a cost-effective answer for solvent drying. Their balance at high temperatures and compatibility with an extensive variety of solvents further decorate their application in numerous business packages.
2. How long does it commonly take to dry a solvent with the usage of molecular sieves?
The drying time can vary depending on several elements, inclusive of the type of solvent, the quantity of water gift, and the unique type of molecular sieve used. Typically, the drying process can take anywhere from some hours to a single day. In a few cases, in particular with solvents which might be more prone to moisture absorption or have higher preliminary water content, the technique might also take longer. For top-of-the-line outcomes, it’s far more important to monitor the drying system intently and conduct periodic moisture tests to decide when the solvent has reached the desired level of dryness.
3. Can molecular sieves be regenerated, and if so, how?
Yes, molecular sieves can be regenerated for reuse, making them a cheap alternative for solvent drying. Regeneration entails heating the sieves to a high temperature, generally around 200-300°C (392-572°F), to take away the adsorbed moisture. The genuine temperature and length depend upon the kind of molecular sieve and the amount of water it has absorbed. After heating, the sieves should be allowed to cool in a dry environment earlier than being stored or reused. This regeneration procedure can be repeated in more than one instance, even though the efficiency of the sieves may steadily lower with every cycle because of potential structural degradation.
4. Are there solvents that can’t be dried by the usage of molecular sieves?
While molecular sieves are noticeably effective for drying an extensive range of solvents, there are some exceptions. For instance, solvents with very low boiling factors or the ones that might be rather reactive with the sieve cloth won’t be suitable for drying with molecular sieves. Additionally, solvents that can be extremely viscous or have massive molecular sizes might not engage correctly with the sieves’ pores, leading to inefficient drying. In such instances, alternative drying techniques or exclusive adsorbents may be important. It is continually advocated to talk with an expert to decide on the first-rate drying method for specific solvents.
5. What is the distinction between exceptional styles of molecular sieves, together with 3A, 4A, and 5A?
The number one difference among 3A, 4A, and 5A molecular sieves lies in their pore length, which determines the size of the molecules they can adsorb. A 3A molecular sieve has a pore length of about 3 angstroms, making it suitable for drying unsaturated hydrocarbons like acetylene and butadiene. A 4A molecular sieve, with a pore length of 4 angstroms, is extra flexible and is generally used for drying gases, alcohols, and preferred solvents. The 5A sieve has a slightly large pore size of five angstroms and is typically used for greater particular programs, which include the removal of carbon dioxide or large molecules from combinations. Selecting the perfect kind of molecular sieve is essential for reaching the surest drying outcomes because it without delay impacts the sieve’s capacity to adsorb moisture without affecting the solvent itself.
Key Takeaways
- Molecular sieves offer precise, reusable solvent drying outperforming silica gel or salts.
- 3A, 4A, and 5A sieves differ by pore size choose based on solvent and molecule size.
- Effective drying requires pre-filtering, proper sieve-to-solvent ratios, and adequate contact time.
- Karl Fischer titration is the best method to verify solvent dryness (<0.01% water).
- Sieves must be regenerated (250°C heat) and stored airtight to remain effective.
- Poor storage, wrong sieve type, or underactivation are the most common failures.
Related Blog Post


Which NDE Methods Apply to Surge Tanks?
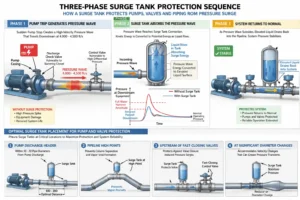
How Do Surge Tanks Protect Pumps and Valves?
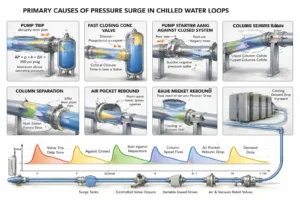
What Causes Pressure Surge in Cooling Loops?
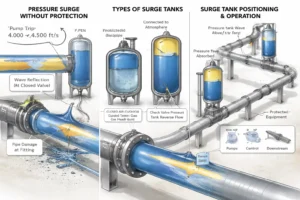
Surge Tanks for Industrial and Process Systems
About Author