Pressure vessels for pharmaceutical manufacturing are essential for sterile, compliant, and efficient drug production. They support critical processes such as mixing, storage, synthesis, and sterilization while meeting strict regulatory standards.
The Importance of Pressure Vessels in Pharmaceutical Manufacturing Depends On
Pressure vessels pharmaceutical manufacturing depend on are foundational to modern drug production. Pharmaceutical environments require absolute control over pressure, temperature, and cleanliness, making specialized pressure vessels essential throughout the manufacturing lifecycle.
In pharmaceutical operations, pressure vessels safely contain liquids and gases under controlled conditions during formulation, chemical reactions, and purification steps. These vessels help manufacturers maintain batch consistency while protecting sensitive compounds from contamination or degradation. Without pressure vessels, pharmaceutical manufacturing would struggle to meet regulatory expectations for sterility and product integrity.
Precision is especially critical when producing injectables, biologics, and vaccines. Pressure vessels pharmaceutical manufacturing facilities use are designed with hygienic finishes, smooth welds, and cleanable surfaces to support sterile environments and validated processes.
Design and Regulatory Standards in Pharmaceutical Manufacturing
ASME and BPE Compliance
Pressure vessels pharmaceutical manufacturing operations use must comply with rigorous standards. ASME Section VIII governs pressure integrity, while ASME BPE focuses on hygienic design and cleanability for bioprocessing applications. Together, these standards ensure vessels are structurally sound and suitable for sterile production environments.
Material Selection for Clean Processing
Most pharmaceutical pressure vessels are fabricated from 316L stainless steel. This material resists corrosion, tolerates repeated sterilization cycles, and supports electropolished finishes. Smooth internal surfaces reduce microbial adhesion and simplify cleaning validation.
Cleanability and Automation
Pressure vessels and pharmaceutical manufacturing systems rely on automated cleaning technologies. Clean-in-Place and Steam-in-Place systems allow vessels to be cleaned and sterilized without disassembly, reducing downtime and limiting operator exposure. These features improve efficiency while supporting consistent sanitation.
Pharmaceutical Processes That Use Pressure Vessels
Biologics and Cell Culture Applications
Biologic drug production depends heavily on pressure vessels. Bioreactors and fermenters maintain controlled pressure, oxygen levels, and temperature to support cell growth and protein expression. Even minor deviations can compromise yield, making vessel performance critical.
Integrated sensors allow real-time monitoring of pressure and process variables. This ensures biologics manufacturing remains stable and repeatable across batches.
Liquid and Semi-Solid Drug Formulation
Pressure vessels that pharmaceutical manufacturing teams use are essential for producing syrups, suspensions, creams, and injectable solutions. These vessels enable controlled mixing, heating, cooling, and homogenization under pressure.
Many formulation vessels are jacketed to manage temperature precisely. Vacuum capabilities help remove entrapped air, improving product stability and shelf life.
Ingredient Storage and Transfer
Active pharmaceutical ingredients are often sensitive to air or moisture. Pressure vessels provide sealed storage environments with inert gas blanketing to preserve material integrity. These vessels also support closed transfer systems that minimize contamination risk during movement between process stages.
Chemical Synthesis and Reactor Vessels
Reactor vessels are a specialized category of pressure vessels that pharmaceutical manufacturing relies on for chemical synthesis. They support controlled reactions through precise regulation of pressure, agitation, and temperature.
These vessels enable repeatable production by supporting pH control, sampling, and heat exchange during synthesis. Their reliability directly impacts product quality and yield.
Final Filtration and Sterilization
Before packaging, many pharmaceutical products undergo final filtration or sterilization. Pressure vessels provide the force needed to push solutions through fine filters while maintaining sterile conditions. Any failure at this stage can compromise an entire batch.
The Role of Pressure Vessels in Pharmaceutical Manufacturing Relies On
The pressure vessels in the pharmaceutical manufacturing sector depend on support for sterility, scalability, and compliance. These vessels enable manufacturers to meet increasing demand for biologics, vaccines, and advanced therapies without sacrificing safety or quality.
As pharmaceutical production grows more complex, pressure vessels continue to evolve with improved materials, smarter monitoring, and greater automation. They remain essential infrastructure for reliable drug manufacturing.
Need a reliable partner?
Red River specializes in the design and manufacturing of pressure vessels. We also fabricate related items such as prefabricated spools and skid packages.
Learn more through our contact page, submit a project via our request a quote, or explore related solutions, including pressure vessels, custom fabrication, and skid packages.
Frequently Asked Questions
1. What are pressure vessels used for in pharmaceutical manufacturing?
Pressure vessels pharmaceutical manufacturing rely on are used for mixing, storing, reacting, filtering, and transferring liquids and gases under controlled pressure and sterile conditions.
2. Which standards govern pressure vessels in pharmaceutical manufacturing?
Pressure vessels in pharmaceutical manufacturing facilities typically follow ASME Section VIII for pressure integrity and ASME BPE standards for hygienic and sanitary design.
3. Why is stainless steel preferred in pharmaceutical pressure vessels?
316L stainless steel is commonly used in pressure vessels. Pharmaceutical manufacturing depends on it because it resists corrosion, supports repeated sterilization, and allows polished finishes that reduce contamination risk.
4. How do CIP and SIP systems support pharmaceutical manufacturing compliance?
Clean-in-Place and Steam-in-Place systems allow pressure vessels used in pharmaceutical manufacturing operations to be cleaned and sterilized without disassembly, reducing contamination risk and downtime.
5. Can pressure vessels be customized for pharmaceutical manufacturing processes?
Yes. Pressure vessels pharmaceutical manufacturing rely on are custom-designed to meet specific pressure, temperature, and cleanliness requirements for regulated drug production.
Key Takeaways
- Pressure vessels for pharmaceutical manufacturing are essential for sterile and compliant drug production
- ASME and BPE standards guide safe and hygienic vessel design
- Stainless steel construction supports durability and cleanability
- Pressure vessels enable biologics, formulation, synthesis, and filtration processes
- Custom designs improve efficiency, safety, and product consistency
Related Blog Post
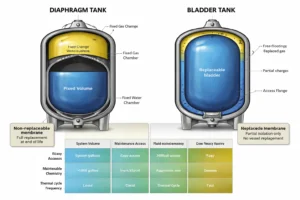
When to Choose Bladder vs Diaphragm Tanks
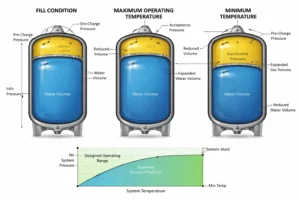
How Do Expansion Tanks Control Loop Pressure?

Expansion Tanks for Data Center HVAC

Which Tests Verify Buffer Tank Quality?

What Size Buffer Tank for Data Centers?
- Pressure Vessels in Manufacturing: Applications and Compliance
- Pressure Vessels in America: Safe & Efficient Chemical Processing
- Pressure Vessels Automotive Industry
- Pressure Vessels Aerospace Industry: Engineering Safety
- Pressure Vessel Supply Chain Challenges in America
- Pressure Vessel Cost Analysis in America’s Industrial Sector